Technology
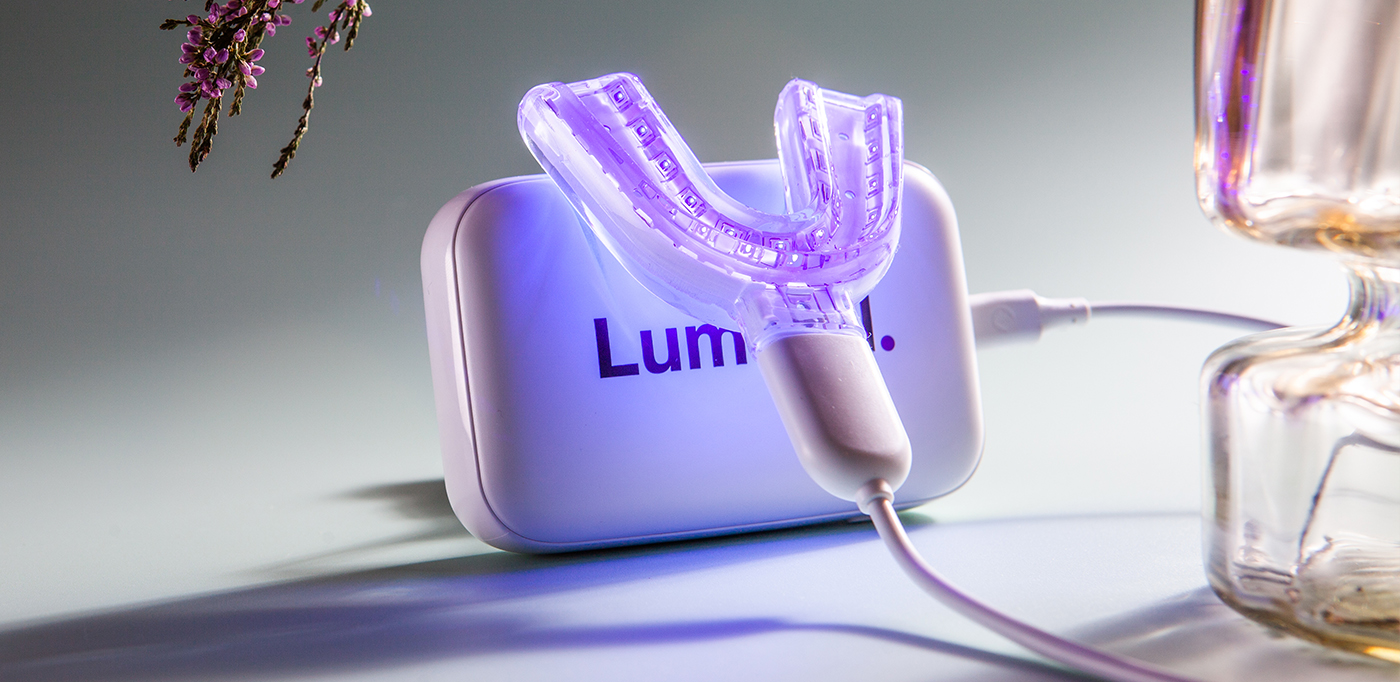
Koite Health’s technology is an effective solution against bacteria for long term use. It is safe, effective and there is no antibiotic resistance formation. Koite Health’s Lumoral is a CE-marked medical device.
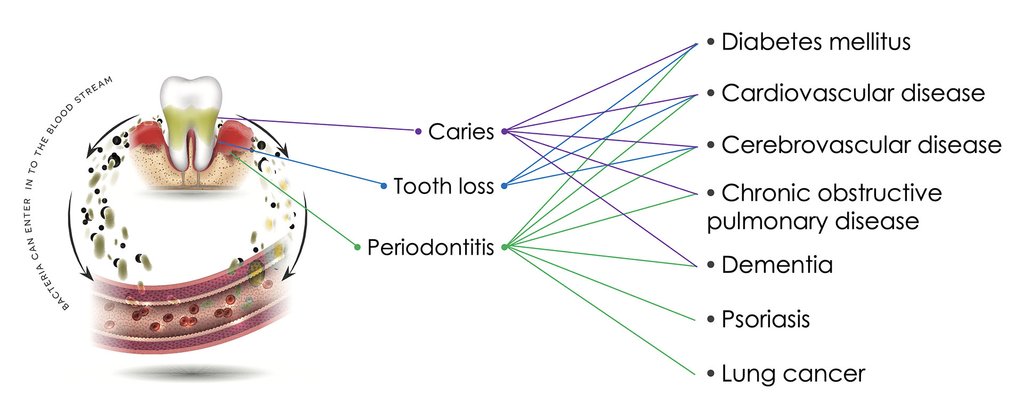
Oral health is a cornerstone of good health. Most of the oral diseases are caused by bacteria.
Caries
Over 60-90% of school-age children and nearly 100% of adults have or have had dental decay
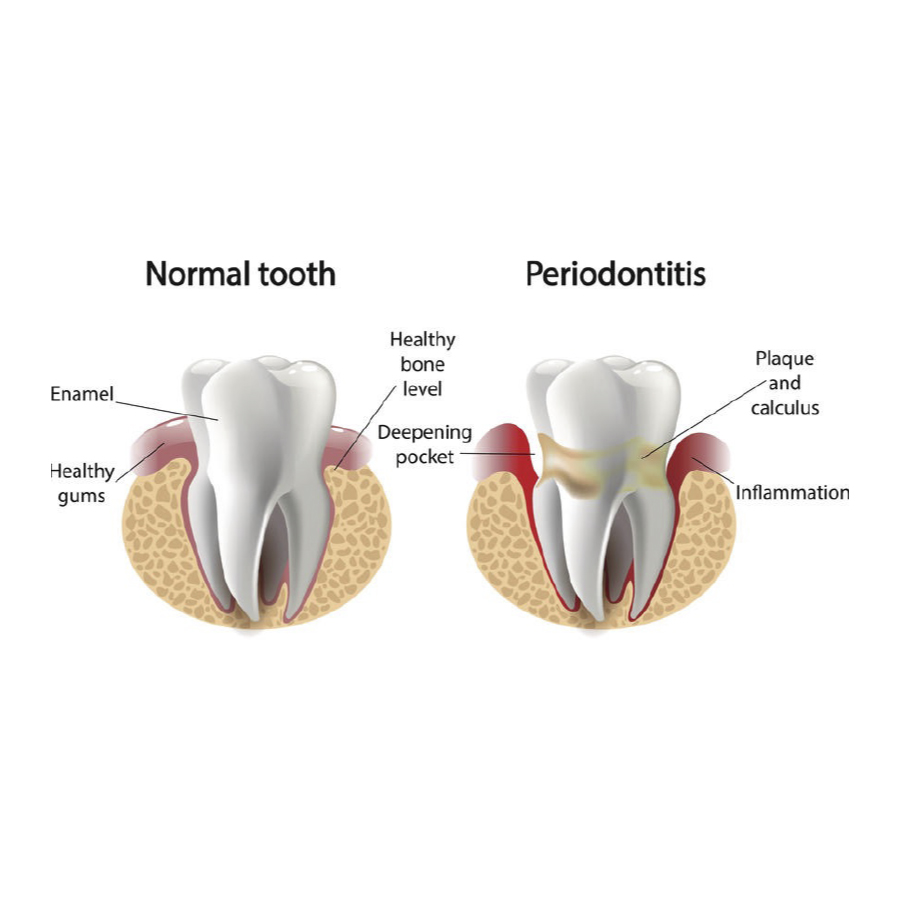
PERIODONTITIS
Around the world, the occurrence of severe periodontitis (gum disease) is 11.2 %, which has remained at the same level since 1990.
Gingivitis
74% of adults suffer from gingivitis, and 21% have deep gum pockets.
PERI-IMPLANT MUCOSITIS
Peri-implant mucositis occurs in 43% of patients after surgery. And, more severe peri-implantitis occurs in 22% of patients post-surgery.